Role of Hyperbaric oxygen therapy in Osteoporosis
Osteoporosis
The most prevalent metabolic bone ailment, osteoporosis, which affects 200 million people globally, continues to be a significant issue. Because osteoporosis is a condition that is clinically silent until it manifests as a fracture, it is frequently undertreated and underrecognized.
Osteoporosis, a metabolic bone disease characterized by poor bone density and degeneration of bone architecture that raises the risk of fractures, affects over 10 million of men and women in the United States. Osteoporosis-related fractures can raise expenditures for overall health care, cause disability, necessitate nursing home placement, and increase mortality [1].
Types
1. Primary Osteoporosis
It is frequently linked to ageing and a lack of sex hormones. Age-related osteoporosis is caused by the trabeculae in bone continuously deteriorating. Additionally, postmenopausal women’s decreased estrogen production significantly accelerates bone loss. In men, sex-hormone–binding globulin inactivates testosterone and estrogen as ageing signs of progress [2].
2. Secondary Osteoporosis
Numerous coexisting conditions and drugs can result in secondary osteoporosis. Osteoporosis-related illnesses frequently include physiological processes including calcium, vitamin D, and sex hormones that are out of balance. For instance, it has been discovered that Cushing’s syndrome accelerates bone loss by producing too many glucocorticoids. In addition, numerous inflammatory conditions, including rheumatoid arthritis, have been linked to secondary osteoporosis and may call for the patient to receive long-term glucocorticoid therapy. Notably, the most often prescribed drugs thought to cause drug-induced osteoporosis are glucocorticoids [3].

Screening and Diagnosis
In general, most organizations advise BMD screening for all persons with a history of fractures who are older than 50. The Preventive Services Task Force advises BMD screening for all women 65 years and older, as well as for younger women with fracture risks that are comparable to or higher than those of Caucasian women 65 years and older who are healthy and have no extra risk factors. The Endocrine Society advises screening all males aged 70 years and older as well as men aged 50 to 69 who have significant secondary osteoporosis risk factors [4].

BMD tests, particularly in the hip and lumbar spine using the dual-energy x-ray absorptiometry (DXA) equipment, or the occurrence of nontraumatic hip or vertebral fractures are used as the gold standard for diagnosing osteoporosis. BMD interpretation and correlation with fracture risk are done using the resulting T-scores. For instance, there is a direct correlation between high fracture risk and low BMD (or a negative T-score).
Another diagnostic tool, named FRAX, was created by the University of Sheffield in Great Britain and is accessible in print or online (Fracture Risk Assessment Tool). The 10-year probability of hip fracture and other major osteoporotic fractures is predicted by considering risk factors like age, race, alcohol use, gender, body mass index, smoking history, prior personal or parental history of fracture, use of glucocorticoids, secondary osteoporosis, rheumatoid arthritis, and femoral neck BMD measurements [5].
Treatments
1) Antiresorptive agents
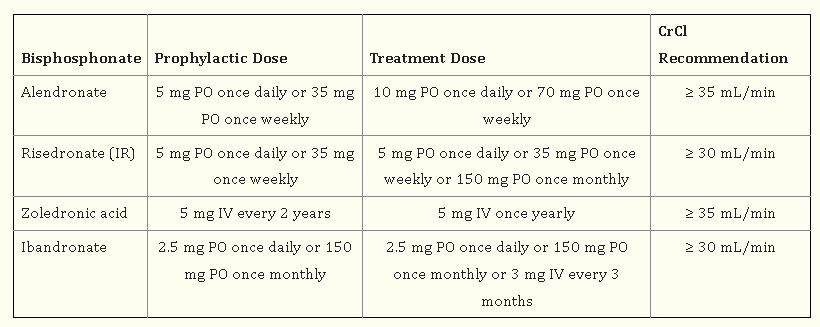
Bisphosphonates and denosumab are used as antiresorptive agents to treat osteoporosis. The prevention and/or treatment of osteoporosis in postmenopausal women, males, and/or GIO patients is advised as the first line of treatment with bisphosphonates, except for ibandronate, by AACE/ACE, ACR, NAMS, and the Endocrine Society. By inhibiting osteoclasts from resorbing bone, bisphosphonates bind to the mineral matrix of the bone with a high affinity, reducing bone turnover and increasing bone mass overall. Denosumab is advised by the AACE/ACE as first-line therapy for individuals who are at substantial risk of fracture and who are unable to take oral medication. The first biologic drug for the treatment of osteoporosis was denosumab. RANKL is inhibited by this completely human monoclonal antibody, which lowers bone resorption. A transmembrane protein called RANKL is necessary for osteoclast development, survival, and operation [6].
2) Hormonal Therapies
A) Estrogen Agonists/Antagonists
An EAA known as raloxifene has both agonistic and antagonistic effects on estrogenic pathways.
a) Raloxifene
Raloxifene increases bone mineral density (BMD) by reducing bone resorption and bone turnover in the bone. Additionally, it exerts estrogen-antagonistic effects on uterine and breast tissue.
b) Conjugated Estrogens/Bazodoxifen
In 2013, the FDA approved the use of a conjugated estrogen and bazedoxifene combination (Duavee, Pfizer) in postmenopausal women with an intact uterus for the treatment of mild to moderate vasomotor symptoms and the prevention of osteoporosis. As an EAA, bazedoxifene lowers the risk of endometrial hyperplasia brought on by the estrogen component [7].
c) Estrogen-Progestin Therapy
In terms of managing osteoporosis, estrogen medication is only FDA-approved for preventing osteoporosis in high-risk postmenopausal women and ought to only be used if other non-estrogenic osteoporotic treatments have been deemed ineffective.
B) Testosterone Therapy
The Endocrine Society advises men at substantial risk of fracture to combine anti-fracture therapy with testosterone therapy despite the paucity of evidence on the use of such combinations. When antiosteoporotic therapy is contraindicated and a person’s serum testosterone level is less than 200 ng/dL, testosterone monotherapy is advised. It is also advised for people who are at borderline substantial risk for fracture, have serum testosterone levels below 200 ng/dL, and exhibit signs or symptoms of androgen deficiency or hypogonadism [9].
C) Calcitonin
With characteristics resembling those of the naturally occurring calcitonin found in humans, birds, and fish, calcitonin is a synthetic polypeptide hormone. When other treatments are not practical, the FDA has approved calcitonin for the treatment of osteoporosis in women who have been postmenopausal for more than five years.
Pathophysiology
Bones give the body structure, safeguard the organs, and store minerals like calcium and phosphorus that are crucial for bone growth and stability. At around 30 years of age, people reach their maximal bone mass. Thereafter, individuals gradually start to lose bone mass. Bones undergo constant remodeling throughout life, which involves the resorption of old bones by osteoclasts & its replacement by newly formed bone produced by osteoblasts. This procedure enables mechanical repair and strength maintenance. The pathophysiological alterations seen in osteoporosis may be caused by an imbalance in remodeling activity where resorption is greater than production.
The main way that estrogen and testosterone affect bone remodeling is by preventing bone breakdown. Additionally, cytokines that affect remodeling have been found, such as nuclear factor kappa-B ligand-receptor activator (RANKL). Osteoblasts that attach to the RANK receptors on osteoclasts create RANKL, which causes the osteoclasts to become activated and mature and result in bone resorption. A strong protease known as cathepsin K has been discovered thanks to recent developments in molecular bone biology (CatK). Activated osteoclasts release CatK during the process of bone resorption, which causes the breakdown of the bone matrix and the mineral components of bone tissue. By indirectly promoting osteoblast proliferation by regulating calcium homeostasis, parathyroid hormone (PTH) has a significant impact on bone formation [10].
Hyperbaric Oxygen as Effective Medicine for Osteoporosis
Hyperbaric oxygen therapy (HBOT) raises the amounts of oxygen in the blood and tissues (Hyperoxia) by delivering pure oxygen at high pressure (about 2-3 atmospheres). Hyperbaric oxygen therapy (HBOT), a type of treatment, involves being exposed to pure oxygen (O2) concentrations at high atmospheric pressures. This pressure may be greater than or equivalent to 1.4 atmospheres (atm), according to the Undersea & Hyperbaric Medical Society (UHMS). Patients must only breathe oxygen while confined in an area with a minimum air pressure of 2 ATA for all current UHMS-approved uses. There must be a pressure environment that is higher than the air pressure at sea level for hyperbaric medicine to be effective. For hyperbaric oxygen therapy, a 100% oxygen delivery system and pressure chamber—which might be flexible or rigid—are necessary. Trained specialists monitor the patient and adhere to a predefined strategy, modifying it, as necessary.
According to research, hyperbaric oxygen therapy (HBOT) can revive bone cells and promote natural growth. HBOT encourages the formation of collagen, which is used to fill in the gaps between shattered bones so they can heal. The development of osteoblasts and osteoclasts, which are essential for removing old, dead bone and building new bone, may also be increased by hyperbaric oxygen. In some situations, patients who had treatments regularly over time saw pain alleviation and even started to restore their range of motion.
In a case study, a 63-year-old woman with post-menopausal osteoporosis is treated with HBOT. After receiving HBOT, there was a clear sign of reduced osteoporosis. For four weeks, the patient received 20 daily sessions [11].
It has been demonstrated that hyperbaric oxygen therapy boosts the production of collagen, a substance that heals the spaces between damaged bones. HBOT also promotes capillary development in the afflicted areas, which aids in supplying the bones with nutrition. The development of new blood vessels is facilitated by hyperbaric oxygen, which facilitates the delivery of oxygen and other nutrients to the bones. The development of osteoblasts and osteoclasts, which are essential for removing old, dead bone and building new bone, may also be increased by hyperbaric oxygen [12].
Hyperbaric therapy in a single-chamber setting. The patient was aware of the importance that hyperbaric oxygen therapy played in research.in osteoporosis treatment before beginning therapy. Each session lasted 90 minutes and included hyperbaric at 2.0 ATA for 90 minutes [13]. Following treatment, the patient’s serum CTx values were 33% lower, demonstrating a general decline in bone resorption. Further evidence for this is that serum bone ALP levels dropped, and lumbar DXA scans showed a slight increase in bone mineral density.
The actions of certain antibiotics within the bone tissue may potentially be stimulated by the elevated oxygen level. The normal treatment cycle lasts for four to more weeks and consists of two hours per day, five days per week. But for patients with persistent wounds and osteomyelitis risk, the time investment is worthwhile [14].
Researchers recently demonstrated that a dynamic oxygen environment is essential for effective bone regeneration associated with an epimorphic regenerative response, considering oxygen’s long-standing role in both bone growth and repair [15].
To promote healing, HBOT is used. The body obtains higher levels of oxygen into the tissue when 100% oxygen is administered. A 2007 study that appeared in Connective Tissue Research demonstrated that HBOT treatments promoted osteoblast formation. Hyperbaric oxygen therapy efficiently cures bone breaks and fractures, according to a study [16].
In a 2013 study, HBOT for osteomyelitis in vulnerable hosts is examined and published in The Open Orthopaedics Journal. According to research, HBOT treatments are a significant treatment option in addition to standard therapies like surgical debridement and antibiotic therapy for patients at higher risk [17]. Researchers examined the effects of high oxygen concentrations in HBTO on the expression of genes involved in bone metabolism in osteoblastic cell lines and human bone in a study. In the event of repeated HBTO sessions (2-3 Atmospheric), the expression of several genes connected to bone metabolism was raised [18].
People in their middle years can develop transitory osteoporosis of the hip (TOH), a rare illness. People with TOH have a gradually increasing level of pain along with a decrease in their range of motion. One month after giving birth, a 33-year-old woman with acute hip pain was investigated in a case study by the department of underwater and hyperbaric medicine. An MRI of the patient revealed a severe bone injury. She underwent a total of 30 sessions of hyperbaric oxygen therapy (2 hours at 2 ATA). She experienced pain relief during her treatment, and after a month, she stopped experiencing any symptoms. She also discovered that her range of motion had fully returned. Following hyperbaric oxygen therapy, an MRI revealed that her bone structure was normal [19].
References
[1]. Salmón-González, Z., et al. “Influencia del oxígeno an alta concentración en cámara hiperbárica sobre el metabolismo óseo.” Revista de Osteoporosis y Metabolismo Mineral 12.1 (2020): 28-31.DOI: http://dx.doi.org/10.4321/S1889-836X2020000100005
[2]. Wright N, Looker A, Saag K, et al. The recent prevalence of osteoporosis and low bone mass in the United States based on bone mineral density at the femoral neck or lumbar spine. J Bone Miner Res. 2014;29(11):2520–2526
Link: https://asbmr.onlinelibrary.wiley.com/doi/full/10.1002/jbmr.2269
[3]. Lin, Julie T., and Joseph M. Lane. “Osteoporosis: a review.” Clinical Orthopaedics and Related Research (1976-2007) 425 (2004): 126-134.
[4]. Raisz LG. Pathogenesis of osteoporosis: concepts, conflicts, and prospects. Journal Clin Invest. 2005;115(12):3318–3325.
DOI: 10.1172/JCI27071
Link: https://www.jci.org/articles/view/27071
[5]. Jeremiah MP, Unwin BK, Greenawald MH, et al. Diagnosis and management of osteoporosis. Am Fam Physician. 2015;92(4):261–268.
[6]. Das S, Crockett J. Osteoporosis—a current view of pharmacological prevention and treatment. Drug Des Devel Ther. 2013; 7:435–448.
DOI: https://doi.org/10.1016/S0140-6736(10)62349-5
Link: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5768298/
[7]. Cosman F, de Beur S, LeBoff M, et al. Clinician’s guide to prevention and treatment of osteoporosis. Osteoporos Int. 2014;25(10):2359–2381. Erratum in: Osteoporos Int 2015;26(7)2045–2047.
Link: https://pubmed.ncbi.nlm.nih.gov/25182228/
[8]. Rossouw JE, Anderson GL, Prentice RL, et al. Risks and benefits of estrogen plus progestin in healthy postmenopausal women: principal results from the Women’s Health Initiative randomized controlled trial. JAMA. 2002;288(3):321–333.
Link: https://pubmed.ncbi.nlm.nih.gov/12117397/
[9]. Watts NB, Adler RA, Bilezikian JP, et al. Osteoporosis in men: an Endocrine Society clinical practice guideline. J Clin Endocrinol and Metab. 2012;97(6):1802–1822.
Link: https://pubmed.ncbi.nlm.nih.gov/22675062/
[10]. Buckley L, Guyatt G, Fink HA, et al. 2017 American College of Rheumatology guideline for the prevention and treatment of glucocorticoid- induced osteoporosis. Arthritis Care Res (Hoboken) 2017;69(8):1095–1110. 2ndry
Link: https://pubmed.ncbi.nlm.nih.gov/28585410/
[11]. Qaseem A, Forciea MA, McLean RM, et al. Treatment of low bone density or osteoporosis to prevent fractures in people: a clinical practice guideline update from the American College of Physicians. Ann Intern Med. 2017:1–27.
[12]. Camacho PM, Petak SM, Binkley N, et al. American Association of Clinical Endocrinologists and American College of Endocrinology: clinical practice guidelines for the diagnosis and treatment of postmenopausal osteoporosis—2016. Endocr Pract. 2016;22(suppl 4): S1–S42.
Link: https://pubmed.ncbi.nlm.nih.gov/27662240/
[13]. Clinical Orthopaedics and Related Research (1976-2007): August 2004 – Volume 425 – Issue – pp 126-134
doi: 10.1097/01.blo.0000132404. 30139.f2
[14]. Yagishita K, Jinno T, Koga D, et al. Transient osteoporosis of the hip treated with hyperbaric oxygen therapy: a case series. Undersea & Hyperbaric Medicine: Journal of the Undersea and Hyperbaric Medical Society, Inc. 2016 Nov-Dec;43(7):847-854. PMID: 28777523.
Link: https://europepmc.org/article/med/28777523
[15]. Al Hadi H, Smerdon GR, Fox SW. Hyperbaric oxygen therapy accelerates osteoblast differentiation and promotes bone formation. J Dent. 2015; 43(3):382-8.
https://doi.org/10.1016/j.jdent.2014.10.006
Link: https://www.sciencedirect.com/science/article/pii/S0300571214002802
[16]. Utting JC, Flanagan AM, Brandao-Burch A, Orriss IR, Arnett TR. Hypoxia stimulates osteoclast formation from human peripheral blood. Cell Biochem Funct. 2010;28(5):374-80.
https://doi.org/10.1002/cbf.1660
Link: https://onlinelibrary.wiley.com/doi/abs/10.1002/cbf.1660
[17]. [Alan A. Katz M.D, Gurvinder Singh B.S., and Kristin Thomson DHSc. PA-C (2020); EFFECTIVENESS OF HYPERBARIC OXYGEN THERAPY FOR TREATMENT OF POSTMENOPAUSAL OSTEOPOROSIS -A CASE REPORT Int. J. of Adv. Res. 8 (Jul). 1535-1537] (ISSN 2320-5407)
DOI: 10.21474/IJAR01/11421
[18]. Fernando WA, Leininger E, Simkin J, Li N, Malcom CA, Sathyamoorthi S, et al. Wound healing and blastema formation in regenerating digit tips of adult mice. Developmental biology. 2011;350(2):301–10.
DOI: 10.1016/j.ydbio.2010.11.035
[19]. Sammarco MC, Simkin J, Fassler D, Cammack AJ, Wilson A, Van Meter K, et al. Endogenous bone regeneration is dependent upon a dynamic oxygen event. Journal of bone and mineral research: the official journal of the American Society for Bone and Mineral Research. 2014;29(11):2336–45. 10.1002/jbmr.2261.